
What is Registration?
Registration has long been an integral part of drug discovery, where it plays a crucial role in finding and characterizing new drugs. In registration, we define the identity of substances of interest by evaluating their key characteristics to determine their uniqueness and capture critical IP. For example, the identity of a small organic molecule could be determined by assessing its 2D chemical structure, while a biologic, such as a recombinant protein, could have its identity determined by comparing its amino acid sequence with sequences of other known proteins.
With biologics becoming increasingly important, the ability to keep up with the fluid nature of evolving formats is a further key consideration. This is critical for any modern registration system.
In most cases, we assign corporate identifiers or labels to enable substance identification and tracking as they undergo further characterization and evaluation.
Registration supports many important business processes across discovery, development, production and manufacturing, enabling scientists and managers to:
- Capture knowledge and protect substance IP
- Leverage libraries after purchase or acquisition more rapidly
- Improve collaboration by proving a consistent, definitive record of substances to reduce confusion and streamline handoff time between individuals and teams
- Understand when a molecule was made and by whom
- Track both real entities and those made by computational processes involving artificial intelligence and machine learning (as typified by BIOVIA’s Generative Therapeutics Design)
- Determine whether a substance is available and, if not, where it can be obtained.
BIOVIA has a long history of providing registration solutions, starting in the 1990’s with ISIS and RS3 before moving onto Accord, Isentris and more recently with Chemical Registration and Biological Registration, the latter the first commercially available biological registration system. With Materials Registration on the 3DEXPERIENCE platform, BIOVIA is bringing registration up to date with new capabilities for a new decade of discovery.
Universal Substance Registration
Materials Registration is a cloud-based, universal registration system that handles almost any type of substance. Unlike the vast majority of systems available today that tend to provide dedicated systems for separate user communities of biologists and chemists, Materials Registration offers a single common user experience for use by everyone. This brings many benefits:
- Chemists and biologists only need to learn how to use a single system
- Artificial barriers between chemists, biologists and their data, erected only by having to use separate dedicated systems for chemistry or biology are broken down and removed, enabling them to share information and work together more effectively
- Potential duplication of substance data is much reduced or eliminated
- The work and costs associated with managing and configuring two largely parallel systems is eliminated
- The use of custom code to enable communication between separate registration systems, often from different vendors is no longer required
- It becomes easier to infer, uncover and establish relationships between different substances, even if the relationship spans different substance types (such as between the small molecule and antibody components found in an antibody- drug conjugate or ADC)
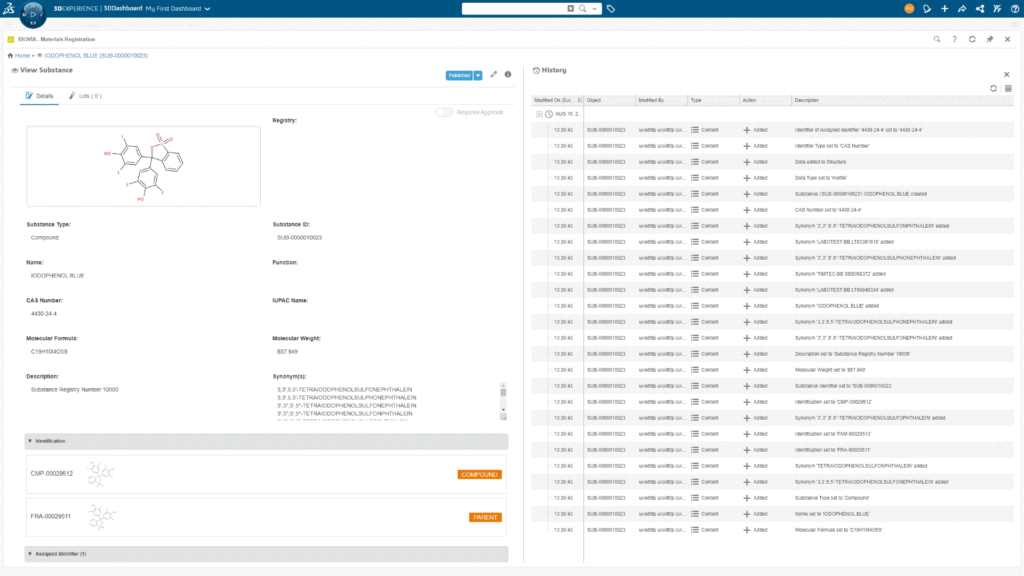
Identity in a Multiple Substance-Type World
An impactful innovation related to offering a single system is the unambiguous way Materials Registration from BIOVIA handles the determination of substance identity. In current systems, the rules used to determine identity typically work against specific substance types. This approach tends to create extra work and duplication in a single system, as you would need to create or assign the same rules to multiple substance types. It also means that you would miss some instances of substance duplication simply because of classification choices made at registration time by scientists (because some systems will only look for duplicates within the same substance type or entity class).
Materials Registration from BIOVIA avoids these limitations by providing identification rules that apply automatically across any substance types with the identifying characteristics they operate on. For example, imagine a scientist registering a single domain antibody as an antibody substance. Then later on, another scientist reports the same substance, classifying it this time as a protein. In Materials Registration, since both substances share the same amino acid sequences, the system will apply the same identification rules to both molecules and recognize that they are identical. Depending on the option selected by the user, the system will either prevent registration of the second substance or assign a shared identity to both. This occurs even though the submitting users have classified them as different substance types.
Multiple Identities
Finally, for today, let us discuss multiple identities in Materials Registration. Until now, traditional registration systems have typically only ever assigned a single identity to a substance, usually a corporate identification label of some kind.
Materials Registration will still allow you to assign a label or ID based on key characteristics such as sequences or structures but it will in addition, allow you to calculate and assign additional labels based on other substance characteristics. For example, by default, Materials Registration will give an identification to a chemical compound based on its chemical structure, but it will in addition assign a secondary label based on a fragment identification rule designed to identify compound “parents”. From this information, it becomes easy to view compounds in families or groups that all share the same common parent fragment, a valuable way to view substances when working on SAR analysis.
Get Involved
Materials Registration builds on the rich heritage of registration at BIOVIA, providing a unified system capable of registering almost any substance. It breaks down barriers between scientists, enables effective collaboration and reduces the cost and complexity involved in supporting parallel systems for chemistry and biology.
If you would like to learn more and get involved with ongoing registration development, I would love to hear from you! Contact me at neil.eccles@3ds.com or sign up and visit the BIOVIA Drug Discovery and Development Community where you will find posts that provide additional information on other aspects of Materials Registration including its role in the wider Materials Management solution underway at BIOVIA!

