Understanding Proton Transfer
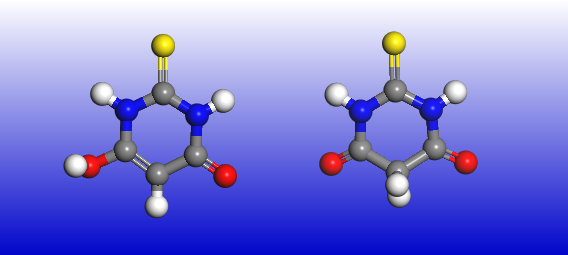
Proton transfer in molecules, both in the gas phase and in the solid state, can proceed quite quickly with a low energy barrier and a small energy difference between the initial and final states. As a result, numerous molecules can exist as different tautomers – an example picture above shows the structures of two tautomers of thiobarbituric acid. Such proton transfer can happen either with or without charge redistribution and either with or without reorganization of chemical bonds. The chemical equilibrium determines the population of tautomeric mixtures, so the ratio of possible tautomers depends on temperature, environment (pH), and solvent. The properties of tautomers can differ, so it is essential to know which one we are dealing with.
How common is tautomerism? This question was reopened and addressed in the recent paper in CrystEngComm by researchers from GSK and Durham University.
More than a decade ago Professor Aurora J. Cruz-Cabeza mined the Cambridge Structural Database (CSD) and reported that the cases of tautomerism among recorded structures are very rare. CSD has evolved significantly in the last decade, while the tools for data analysis also became more detailed and thorough. The latest re-examination by Professor Cruz-Cabeza and GSK scientists had access to many more CSD structures, as well as to the drug subset of the CSD, and to the GSK small molecule crystal structure database. The study discovered that the percentage of compounds capable of tautomerism is significantly higher among drug-like molecules (70%) than the CSD-average value of 50% for all compounds. This explains why the study of tautomers is particularly relevant for pharmaceutical development. The project ran at GSK by the Ph.D. student Amy Woods-Ryan (now an investigator at GSK) included data analysis of the structures recorded in the database, and also an investigation of the relative lattice energies of proton transfer polymorphs.
BIOVIA Materials Studio was a useful tool for this project. Experimental structures from CSD and from GSK databases were analyzed with Materials Studio CASTEP to get accurate estimates of relative energies of tautomeric polymorphs from density functional calculations. The findings confirm that solid-state polymorphs fall within the accepted energy range of 1-7 kJ/mol despite significant energy differences in the gas phase. The authors’ conclusion is that “tautomerism is not just a serious consideration in the pharmaceutical industry for medicinal chemists, but for solid state scientists alike.” Drug-like molecules have an increased tendency to contain functional groups capable of tautomerism, as well as to contain both acidic and basic functional groups, thus enabling zwitterion formation. Evidence suggests that both tautomer equilibrium and speciation in solution may have implications for the solid state outcome and polymorphic control in crystallizations.
This work clearly illustrates the power of data analysis combined with molecular modeling in pharmaceutical development. And also important, I had a lot of fun doing this work.
Amy Woods-Ryan
Conclusion and Future Perspectives
In conclusion, tautomerism, the phenomenon of molecules existing in multiple structural forms, plays a significant role in chemistry, particularly in the pharmaceutical industry. Through the integration of data analysis and molecular modeling, researchers are uncovering valuable insights that aid in pharmaceutical development and enhance our understanding of molecular behavior. Learn more about BIOVIA Materials Studio.
