Challenge:
Accurately simulate the full crimping of delicate bioprosthetic heart valves under extreme deformation to ensure safety and real-world reliability before physical testing.
Solution:
Medtronic engineers worked with simulation experts from the SIMULIA Services Hub and their structural simulation technology, Abaqus, to run high-fidelity, iterative crimp simulations that captured complex nonlinear tissue and material behavior.
Benefit:
Full-crimp modeling accelerates design confidence, cuts the number of costly physical builds, strengthens regulatory evidence, and supports safer, faster development of next-generation transcatheter heart valves.
Simulating Precision: How Medtronic Engineers Advance Heart Valve Innovation
SIMULIA Abaqus enables Medtronic engineers to design and test next-generation heart valves with speed, accuracy, and patient-first precision.
Medtronic has always skillfully combined engineering excellence and medical innovation. From the first battery-powered pacemaker to today’s miniaturized, leadless devices (implanted via a vein), the company’s mission has remained constant: Harness technology to improve lives. That same commitment drives its pioneering work in transcatheter heart valve replacement, a procedure that spares patients the need for open-heart surgery by delivering a bioprosthetic valve through a catheter system.

Over 5 million people in the U.S. alone are diagnosed with heart valve disease each year. Left untreated, the disease can lead to heart failure and death. The condition develops when native valve leaflets stiffen from calcification, forcing the heart to work harder to pump blood. For decades, the only treatment was open-heart surgery, often a high-risk option, especially for older adult patients.
The Hidden Challenge Inside Every Valve
Over the past 20 years, transcatheter heart valve replacement has transformed cardiac care. Instead of opening the chest, surgeons now guide a bioprosthetic valve through a small incision in the groin and deploy it inside the failing native valve. “The bioprosthetic valve is then deployed using the delivery system … and the patient is typically discharged the same day,” explains David Martin, Senior Principal Engineer at Medtronic.
Developing these life-saving devices presents formidable design challenges. Each valve must be crimped to a fraction of its standard size to fit inside a catheter and navigate the body’s vascular system. “The valves could be 20 to 40 mm in diameter,” Martin notes. “To be delivered into the heart, they must be crimped to 6-7mm diameter and loaded into a catheter delivery system. The catheter is then inserted into the femoral artery in the groin and tracked through the aorta into the chambers of the heart”.

The crimping process induces extreme stress and deformation across multiple materials—metal frames, bioprosthetic soft tissue, and sutures—all of which must function flawlessly once deployed. “The focus of our work is ensuring the bioprosthetic valve can be delivered accurately to the target anatomy, where it should then function reliably for decades after deployment” emphasizes Martin. Patient safety is job number one at Medtronic, which is why Medtronic engineers utilize only the highest-end simulation tools in their product development process.
Cracking the Crimping Code
To overcome these complex challenges, Medtronic turned to Abaqus, SIMULIA’s Finite Element Analysis (FEA) software. Using simulation, engineers can virtually model the entire life cycle of a heart valve, from crimping to deployment, before a single prototype is built. Medtronic engineers use modeling tools widely in all stages of the design and development of transcatheter heart valves, including to accelerate concept assessment, for detailed design optimization, and in design verification activities prior to regulatory submission.
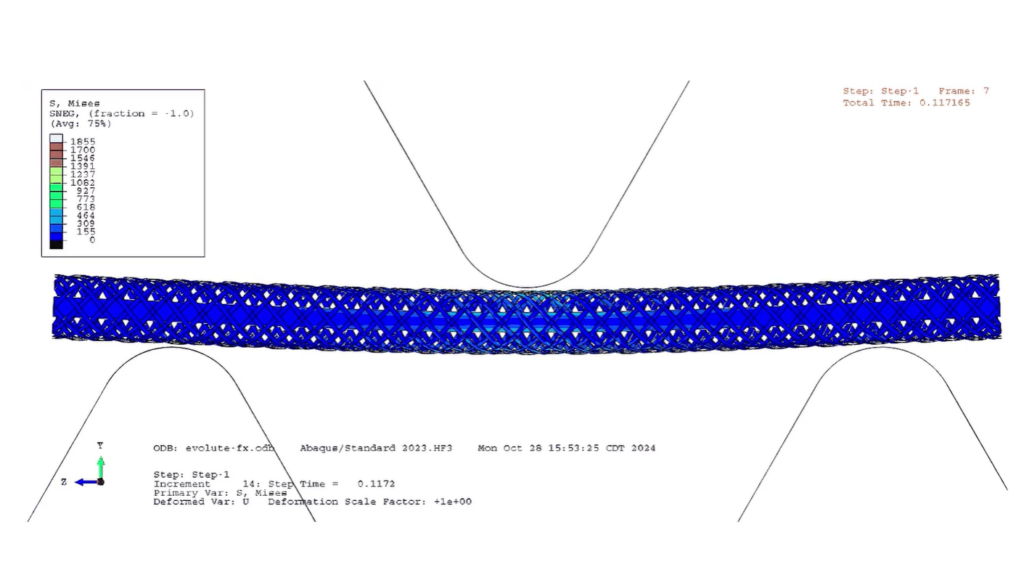
In a recent collaboration, Medtronic engineers teamed up with simulation experts from the SIMULIA Services HUB to tackle one of the toughest steps in heart-valve design: simulating the full crimping process including all the tissue components. “This is a very challenging problem due to the extreme deformations and contact,” Martin says. The challenge wasn’t just compressing the device to fit inside a delivery catheter; it was doing so with the bioprosthetic tissue, treated biological material sourced from animals, attached. Unlike metal or plastic, bioprosthetic tissue stretches, wrinkles, tears, and buckles easily, making the physics particularly unforgiving. “The tissue is incredibly delicate, and the deformations and contact are extreme. There’s a lot of nonlinear materials all coming in contact with one another,” he explains. The work pushes the limits of biomechanical simulation, which ultimately helps engineers build safer valves for patients who rely on them.
SIMULIA Services HUB experts in the UK, worked closely with the Galway Medtronic team over several weeks. “We gave them everything they needed. We contributed where we could, but they carried out the [simulation] work … until they achieved the goals that we agreed in the problem statement,” Martin said. This project not only demonstrated that full-scale crimp simulation could be performed efficiently, but it also provided a deeper understanding of how the tissue components may behave during the crimping process.
Martin credits SIMULIA Abaqus for its proven reliability in such demanding applications: “I’ve personally been using Abaqus because of the long history of good general performance with the solvers.”
Building Confidence Through Simulation
Modeling and simulation have revolutionized the way Medtronic designs, tests, and verifies its devices. Martin clarifies, “Manufacturing and testing the valves that we work on is incredibly costly. Anything we can do upfront to make sure we’re only building and testing valve designs that we are confident in helps us. And that’s where modeling and simulation is really having a big impact in Medtronic.”
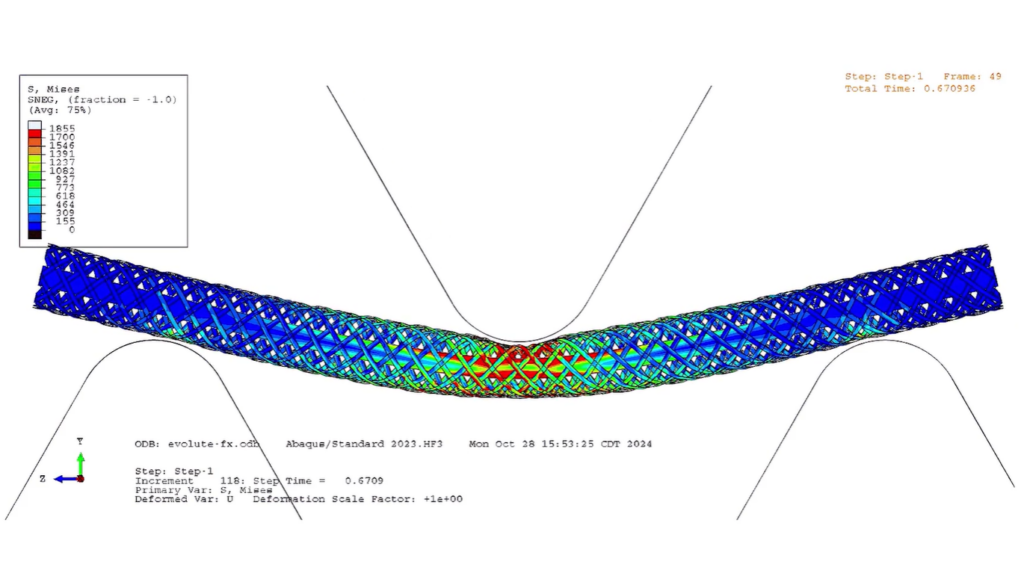
Simulation now supports every stage of development, from early concept exploration to regulatory verification. “We’re going to squeeze every ounce of performance out of it that we can,” Martin adds. The virtual testing data also supports FDA submissions by demonstrating safety and efficacy, which ensures that the device will perform as intended and poses no risk to patients.
In addition, there are substantial efficiency gains. In one project, Medtronic optimized frame designs across 100 analysis runs, work that would have taken months or even years to reproduce physically. (Frame designs are lattice-like components ofmetal stent structures that hold the bioprosthetic tissue in place once the valve is crimped, delivered, and deployed.) “If you consider the time to build and test a hundred frames, that would be months, if not years,” attests Martin. “That’s work that we did … in less than a quarter [three months].” Demonstrating that simulation-driven virtual testing can be done up front and presenting the actual data to the management team makes a strong case for continuing a simulation-driven approach to product development.
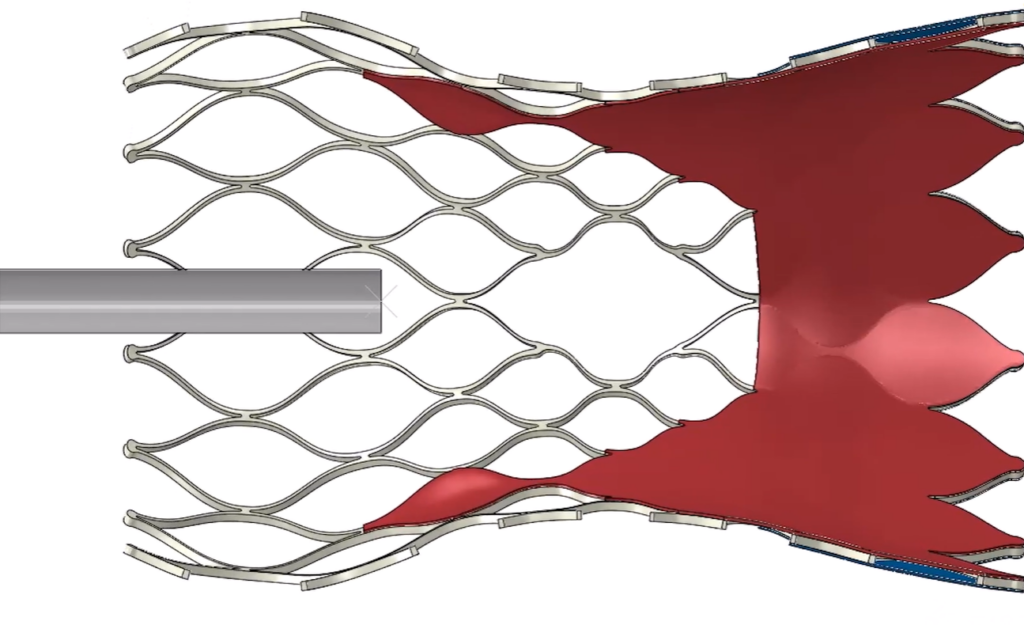
Beyond efficiency, simulation reinforces Medtronic’s uncompromising standards for accuracy and patient safety. “We do a huge amount of work at Medtronic to make sure our models are accurate to real life. Verification and validation establish accuracy,” Martin concludes.
From Validation to Vision
Simulation has become deeply embedded in Medtronic’s engineering culture. “When I joined the company in 2014, there were four dedicated simulation engineers working in the Structural Heart business unit,” Martin recalls. “Now there’s nearly 40 or more.” That tenfold growth underscores the company’s confidence in simulation and its belief that virtual testing is now central to medical device development.
By partnering with SIMULIA and leveraging their structural simulation technology, Abaqus, Medtronic continues to lead the evolution of transcatheter heart valve design while improving reliability, reducing cost and development time, and ultimately delivering safer outcomes for patients worldwide. Once a supporting tool, simulation is now a key cornerstone of how Medtronic turns ideas into life-saving innovations.

Interested in the latest in simulation? Looking for advice and best practices? Want to discuss simulation with fellow users and Dassault Systèmes experts? The SIMULIA Community is the place to find the latest resources for SIMULIA software and to collaborate with other users. The key that unlocks the door of innovative thinking and knowledge building, the SIMULIA Community provides you with the tools you need to expand your knowledge, whenever and wherever.