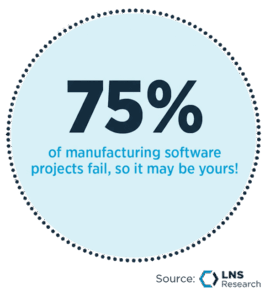
A recent spotlight from LNS Research reveals a stark statistic: 75% of manufacturing operations software initiatives fail.
This figure is alarming. It doesn’t indicate that these programs merely underperform or delay their return on investment. It means they fail completely. Despite the high stakes, many Medical Technology (med-tech) organizations continue to approach Manufacturing Execution Systems (MES) software as a simple plant IT upgrade. They fail to recognize it for what it truly is: a strategic operating model decision.
When leadership views MES as a box-checking exercise or a localized software installation, they miss the broader picture. To succeed, manufacturers must fundamentally rethink how they approach digitization, standardization and global governance. This blog explores why MES programs falter and how a platform strategy can drive transformative success.
MES Is Not Just a Compliance Project
In the medical device industry, leaders often justify MES investments based on regulatory necessities. The business case typically revolves around Device History Records (DHR), Unique Device Identification (UDI) traceability, electronic work instructions and audit readiness.
These elements are undoubtedly critical. However, they aren’t transformational. They represent the baseline requirement for doing business in a regulated environment. If your primary business case relies solely on “we need better compliance,” you’re aiming too low. Compliance keeps the doors open, but it doesn’t drive competitive advantage or operational excellence.
The real challenge med-tech leaders face is scalability. The pertinent question isn’t whether you can pass an audit, but whether your manufacturing system can scale with SKU proliferation. Can you support accelerated New Product Introduction (NPI) and global tech transfer without eroding your margin?
This is the specific problem a modern MES must solve. It needs to function as a mechanism for efficiency and speed, not just a digital filing cabinet for regulatory documents.
The Hidden Risk in Med Tech Manufacturing
The complexity within med-tech manufacturing is exploding. Companies face shorter product lifecycles, a vast increase in variants and configurations, globalized production footprints and a heavy reliance on contract manufacturing partners.
Within this complexity, many companies still run fragmented, plant-specific systems. These legacy systems often rely on heavy customization to fit the unique preferences of a single site.
The consequences of this approach are severe. Organizations experience slow NPI transfer because every site operates differently. Validation costs skyrocket as teams must validate unique software instances for every location. Data models become inconsistent, limiting enterprise visibility and making it impossible to compare performance across sites.
Perhaps most damaging is “upgrade paralysis.” When a system is heavily customized, upgrading to the latest version becomes a risky, expensive and time-consuming project. As a result, systems stagnate on outdated versions, security vulnerabilities increase and leadership wonders why productivity stalls.
The Real Reason MES Programs Fail
It’s rarely the software itself that causes failure. The technology usually works as designed. The failure stems from organizational behavior.
Companies frequently try to automate variability instead of eliminating it. They approach the market with 400-page Requests for Proposals (RFPs) that detail every existing manual process and demand that the digital system replicate them exactly.
This approach allows every site to maintain its “unique” way of doing things. Teams over-customize the software to preserve local habits, effectively paving the cow path. They treat MES as a feature checklist to satisfy current operators rather than an opportunity to redesign the operating model for the future.
When the project inevitably runs over budget, misses timelines and fails to deliver value, stakeholders blame the vendor. In reality, the flaw lay in the strategy to automate chaos rather than standardize processes.
The Hard Truth About Strategy
If your MES strategy is led by individual plants, driven by IT and justified solely by compliance, statistics suggest you’ll fail.
Winning MedTech manufacturers take a different path. They execute three specific strategies differently:
- They standardize before they digitize. They define a common way of working across the enterprise before applying technology.
- They govern globally. Decisions about the manufacturing model happen at an enterprise level, not a local one. Site autonomy is reduced in favor of enterprise agility.
- They measure transformation correctly. Success is measured in margin impact, scrap reduction and release velocity, not just in documentation or “going live.”
These successful organizations build a global Manufacturing Center of Excellence (COE). They deploy global templates that enforce standardization while allowing for controlled configuration. They protect this standardization rigorously. They align their MES implementation to high-level Key Performance Indicators (KPIs) like right-first-time metrics and release velocity.
Why Platform Strategy Matters Now
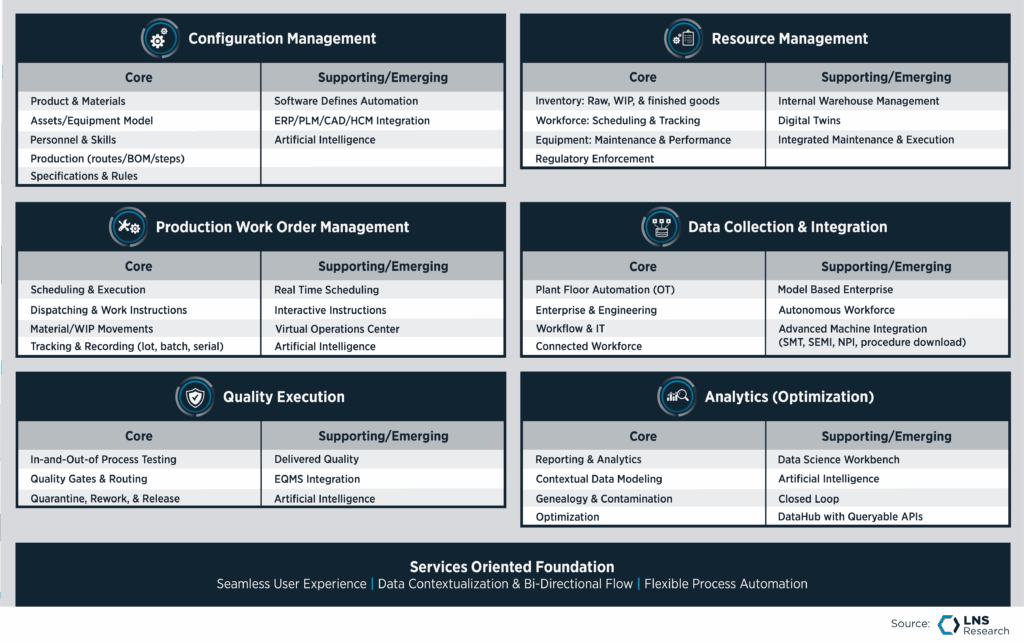
This need for standardization is where enterprise-scale capability becomes decisive. A platform approach connects disparate elements of the product lifecycle into a cohesive whole.
Dassault Systèmes positions itself to solve this by connecting product design and engineering directly with manufacturing process definition, execution systems, quality and traceability.
For MedTech manufacturers, this design-to-manufacturing continuity is no longer optional. Rapid NPI and global tech transfer demand a connected thread of data. When engineering changes a product design, that change must flow directly to the manufacturing floor and the quality checks without manual translation or data reentry.
MES can’t live in isolation anymore. It must exist as part of a broader ecosystem that links the virtual world of design with the physical world of production.
The Leadership Question
The fact that 75% of MES initiatives fail shouldn’t cause leaders to delay investment. Digitization is inevitable and necessary. Instead, the statistic should prompt a serious evaluation of strategy.
Leaders must ask themselves tough questions:
- Are we redesigning our operating model or are we just digitalizing legacy habits?
- Are we building enterprise standardization or are we protecting site autonomy?
- Are we driving margin impact or are we just checking compliance boxes?
MedTech leaders who treat MES as a strategic enterprise platform will outpace competitors. They’ll achieve greater agility, better cost control and true scalability. The rest will continue to digitize complexity. And as the market has shown, complexity never scales profitably.
Where to Learn More
Discover more insights by downloading the LNS Research Paper, Unlock the Secrets to Successful Manufacturing Operations Software Implementation and Med-Tech MOM Whitepaper, Unlocking Growth Through Innovative Manufacturing Solutions. Start your journey toward informed decision-making today.
DELMIA, from Dassault Systèmes, enables manufacturers to keep factory operations running smoothly. Powered by the 3DEXPERIENCE platform, our Manufacturing Operations Management (MOM) and Manufacturing Execution Systems (MES) solutions establish a unified digital environment that provides real-time visibility and AI-enhanced control. By connecting the virtual and real worlds, we enable you to streamline complex processes, minimize waste and guarantee quality. Harnessing data-driven insights and intelligent automation allows for optimized production, enhanced adaptability to disruptions and the delivery of sustainable, customer-focused manufacturing performance at scale.

